Anti-FAK Rabbit mAb
Catalog number :AT0375
Non-receptor protein-tyrosine kinase implicated in signaling pathways involved in cell motility, proliferation and apoptosis. Activated by tyrosine-phosphorylation in response to either integrin clustering induced by cell adhesion or antibody cross-linking, or via G-protein coupled receptor (GPCR) occupancy by ligands such as bombesin or lysophosphatidic acid, or via LDL receptor occupancy. Microtubule-induced dephosphorylation at Tyr-397 is crucial for the induction of focal adhesion disassembly. Plays a potential role in oncogenic transformations resulting in increased kinase activity.
- Overview
- Description
- Rabbit monoclonal antibody to FAK
- Reactivity
- Mouse, Rat, Human
- Tested applications
WB : 1/500 - 1/1000. Detects a band of approximately 125 kDa (predicted molecular weight: 119 kDa). IHC-P : 1/50 - 1/100. ICC/IF : 1/100 - 1/200.
- Properties
- Immunogen
Synthetic peptide conjugated to KLH, derived from around amino acids 923-927 (K-V-Y-E-N) of Human FAK (UniProt Q05397)
- Clonality
- Monoclonal, clone number: 6H7
- Isotype
- Rabbit IgG
- Form
- Liquid
pH: 7.40
Preservative: 0.02% Sodium azide
Constituents: 49% PBS, 50% Glycerol, 0.88% Sodium chloride
- Storage instruction
- Store at -20°C. Stable for 12 months at -20°C
- Database links
- Entrez Gene: 5747 Human
- Entrez Gene: 14083 Mouse
- Entrez Gene: 25614 Rat
- Omim: 600758 Human
- SwissProt: Q05397 Human
- SwissProt: P34152 Mouse
- SwissProt: O35346 Rat
- Unigene: 395482 Human
- Applications
- WB Image
 All lanes : Anti-FAK antibody (AT0375) at 1/500 dilution
All lanes : Anti-FAK antibody (AT0375) at 1/500 dilution
Lane 1 : HeLa cell extract
Lane 2 : HeLa cell extract with blocking peptide
Predicted band size : 119 kDa
Observed band size : 125 kDa
Why is the actual band size different from the predicted?
Western blotting is a technique that separates proteins based on size - in general, the smaller the protein the faster it migrates through the gel. However, migration is also affected by other factors and so the actual band size observed may differ from that predicted. Common factors include...
- post-translational modification - e.g. phosphorylation, glycosylation etc which increases the size of the protein
- post-translation cleavage - e.g. many proteins are synthesized as pro-proteins, and then cleaved to give the active form, e.g. pro-caspases
- splice variants - alternative splicing may create different sized proteins from the same gene
- relative charge - the composition of amino acids (charged vs non-charged)
- multimers - e.g. dimerisation of a protein. This is usually prevented in reducing conditions, although strong interactions can result in the appearance of higher bands
- IHC Image
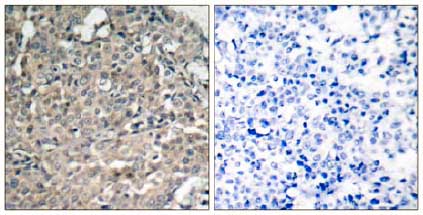 Immunohistochemical analysis of paraffin-embedded Human breast carcinoma tissue labelling FAK with AT0375 at 1/50 dilution. Right panel was preincubated with blocking peptide.
Immunohistochemical analysis of paraffin-embedded Human breast carcinoma tissue labelling FAK with AT0375 at 1/50 dilution. Right panel was preincubated with blocking peptide.
- ICC/IF Image
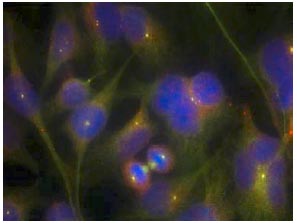 Immunofluorescent analysis of methanol-fixed HeLa cells staining FAK with AT0375 at 1/100 dilution.
Immunofluorescent analysis of methanol-fixed HeLa cells staining FAK with AT0375 at 1/100 dilution.